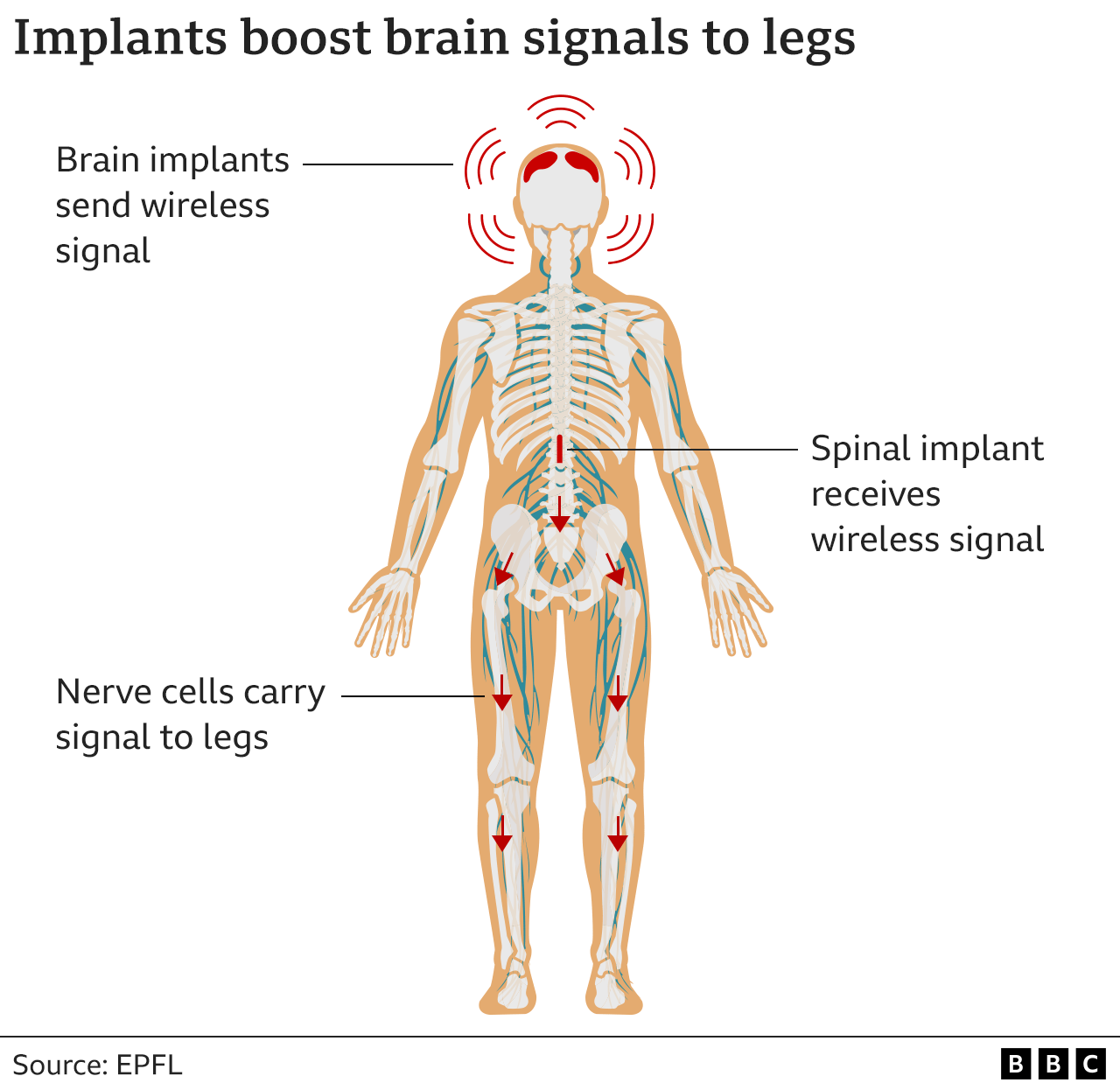
The revolutionary wireless device, which reads brain waves and sends instructions to the spine to move the right muscles, has allowed him to regain his natural mobility simply by thinking about it.
In 2011, Gert-Jan Oskam was in a motorcycle accident that left him paralysed from the waist down. Now, thanks to revolutionary new technology, neuroscientists have given him control over his lower body again.
‘For 12 years I’ve been trying to get back on my feet,’ he said in a press briefing. ‘I have learned how to walk normally, naturally.’
In a study published in the journal Nature, Swiss researchers detailed the functionality of the device, which in short provides a ‘digital bridge’ between Oskam’s brain and spinal column, bypassing any injured sections.
This has allowed him to stand, walk, climb stairs, and ascend a steep ramp with only the assistance of a walker.
Over a year after the implant was inserted, he has retained these abilities, and has actually shown signs of neurological recovery, walking with crutches even when the implant was switched off.

‘What we’ve been able to do is re-establish communication between the brain and the region of the spinal cord that controls leg movement with a digital bridge,’ explained Professor Grégoire Courtine at the Swiss Federal Institute of Technology (EPFL), which runs a longstanding programme to develop brain-machine interfaces to overcome paralysis.
‘The system can capture the thoughts of Gert-Jan and translate those thoughts into stimulation of the spinal cord to re-establish voluntary leg movements.’
The system, which (despite being encouraging) is still at an experimental stage, works by electronically transmitting Oskam’s thoughts to his legs and feet via a second implant in his spine.
Using wireless signals, it reconnects the brain with muscles that are rendered useless when spinal cord nerves are broken.