By recreating the extreme conditions of exoplanets, a team of researchers have successfully managed to produce nanodiamonds from PET plastic.
As the climate disaster worsens, our need for technological solutions becomes more urgent.
Over the last decade, research into climate innovation has produced a mixed bag of results despite the mounting pressure. Some ideas have gained traction and are genuinely viable, while others have proven to be mostly implausible and economically impossible.
However, as our understanding of climate change improves, we are able to observe and discover new ways to curb emissions and – in some cases – even reverse the environmental damage we’ve already caused.
One such example is in Germany, where researchers have found a way to turn PET plastics from water bottles, food packaging, and other containers into nanodiamonds by recreating the extreme conditions of exoplanets. Fancy stuff.
It’s a significant breakthrough that tackles two problems at once. Not only could this transform the way we dispose of plastic waste, but it could also help address ethical concerns surrounding the practice of mining raw materials.

All that’s well and good, of course, but how exactly was this achieved?
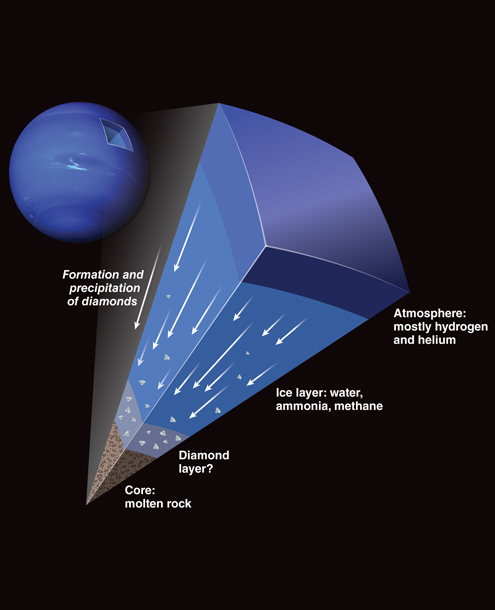
In 2017, the research team set out to replicate specific weather conditions already observed on Neptune and Uranus using polystyrene. Both planets endure intense pressure and high temperatures beneath their surfaces, causing their atmospheres to produce diamonds. Yes, it rains diamonds.